Research compliance portals: Unlocking power for peptide studies

TL;DR:
- Research compliance portals have evolved into centralized systems that safeguard data integrity, accelerate review processes, and improve peptide study management. They offer role-based access, audit trails, automated workflows, and secure documentation to meet complex regulatory frameworks like HIPAA, FDA standards, and GDPR. Embracing advanced, adaptable portals enhances research speed, funding prospects, and data credibility in peptide science.
Research compliance portals have evolved far beyond simple document repositories, and the institutions that recognize this first are pulling ahead in peptide science. Where many investigators still see compliance infrastructure as a layer of bureaucracy standing between the lab bench and meaningful discovery, forward-thinking research teams are treating these platforms as operational command centers that protect data integrity, accelerate review timelines, and position studies for funding. This article breaks down how these portals function, what their built-in mechanics actually deliver in the context of peptide procurement and study management, and where the technology is heading next.
Table of Contents
- What are research compliance portals?
- Core mechanics: Features that drive security and efficiency
- Data privacy, regulatory requirements, and peptide procurement
- Advanced features: Enterprise solutions for high-stakes peptide research
- Edge cases, challenges, and evolving compliance standards
- Our perspective: Compliance portals—asset or obstacle?
- Optimize your peptide research workflow with trusted portals
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Centralized oversight | Compliance portals centralize management, making complex peptide research more secure and efficient. |
| Integrated compliance | Automation connects protocol submissions, tracking, and training into one streamlined system. |
| Enhanced data security | Role-based access, audit trails, and data logging help protect sensitive information and maintain privacy law adherence. |
| Scalable solutions | Enterprise platforms offer advanced risk management and regulatory adaptability for collaborative or high-stakes research. |
| Evolving challenges | Researchers must watch for regulatory changes and edge cases, using portals as active tools to avoid compliance gaps. |
What are research compliance portals?
Research compliance portals are centralized electronic platforms used by universities and research institutions to manage submissions, reviews, and approvals for protocols related to IRB (human subjects), IACUC (animal use), IBC (biosafety), and COI (conflict of interest). Rather than routing paper forms across departments or managing approvals through fragmented email chains, these platforms consolidate every step of the compliance lifecycle into a single, auditable environment.
Understanding the foundations of peptide research makes it immediately clear why this kind of infrastructure matters. Peptide studies often involve sensitive biological materials, cross-departmental teams, vendor relationships, and multi-phase protocols. Without centralized oversight, even a minor documentation gap can stall a study or trigger a regulatory finding.
The core modules typically housed in a compliance portal include:
- IRB (Institutional Review Board): Human subjects protocols, consent forms, and continuing review submissions
- IACUC (Institutional Animal Care and Use Committee): Animal use protocols and welfare certifications
- IBC (Institutional Biosafety Committee): Oversight of biohazardous materials and containment procedures
- COI (Conflict of Interest): Disclosure forms and financial relationship tracking
- Training management: Linkage to mandatory training completion records (e.g., CITI, RCR certifications)
The difference between managing these elements through traditional paperwork versus a portal-based system is significant.
| Dimension | Traditional compliance | Portal-based compliance |
|---|---|---|
| Submission method | Paper or email | Electronic, structured forms |
| Review routing | Manual, departmental handoffs | Automated workflow routing |
| Tracking visibility | Minimal, status by request | Real-time dashboard tracking |
| Document storage | Physical files or scattered folders | Centralized, version-controlled |
| Audit readiness | Labor-intensive retrieval | Instant, searchable records |
| Turnaround time | Weeks to months | Typically 30 to 50% faster |
That last row is not a marginal improvement. For teams managing tirzepatide research compliance and similar high-interest peptide compounds, a multi-week delay in protocol approval can disrupt procurement cycles, affect supplier lead times, and push publication timelines back by entire quarters. The administrative efficiency gains translate directly into research velocity.
These portals also automate workflows including electronic submission and routing for committee review, progress tracking, document storage, training notifications, and annual compliance certifications. For high-volume peptide programs, that level of automation is not a luxury. It is a baseline operational requirement.
![]()
Core mechanics: Features that drive security and efficiency
Understanding what these portals are, it is critical to see how their built-in mechanics deliver real security and operational efficiency, especially under the unique pressures of peptide research. The feature architecture behind most enterprise compliance platforms follows a consistent logic: control access rigorously, capture every action, and integrate with the training and regulatory systems investigators already use.
The standard setup for user roles and access in a research compliance portal follows a clear hierarchy:
- Principal Investigator (PI): Full protocol ownership, initiation rights, and final submission authority
- Co-Investigator (Co-I): Can contribute to protocol sections and review submissions, but cannot finalize independently
- Data Entry / Research Coordinator: Limited input rights, restricted from viewing sensitive financial disclosures or identifiable participant data
- IRB or Committee Reviewer: Read access to full protocols, annotation rights, no edit access to investigator sections
- Compliance Administrator: System-level rights for routing, notifications, and reporting, without research data access
This tiered architecture, which includes role-based access control, audit trails, and electronic signatures, and integration with training systems like CITI for RCR and COI, forms the security backbone of any compliant research environment. Every action taken within the system is logged with a timestamp, user ID, and action type.
Pro Tip: One of the most common and costly mistakes in portal setup is over-assigning administrator rights to research coordinators or junior staff for convenience. This violates the principle of least privilege and can create significant liability during audits. Assign roles based strictly on what each user needs to perform their specific function, and review access permissions annually or whenever staff transitions occur.
To illustrate how these features compare across platform types:
| Feature | Basic portal | Mid-tier portal | Enterprise portal |
|---|---|---|---|
| Role-based access control | Yes | Yes | Yes, with custom roles |
| Electronic signatures | Limited | Full | 21 CFR Part 11 compliant |
| Audit trail depth | Basic logging | Full action history | Timestamped, exportable |
| Training system integration | Manual | CITI linkage | Automated verification |
| Real-time notifications | Email only | In-app and email | Configurable multi-channel |
| API/system integration | None | Limited | ERP, LIMS, HR systems |
For peptide research teams managing secure portal setup, understanding where your current portal falls on this spectrum is the first step toward identifying security gaps. A basic portal may suffice for a single-site observational study. A multi-site peptide clinical study involving identifiable biological samples, international collaborators, and proprietary compound sourcing requires the full enterprise feature set.
The importance of these mechanics becomes clearer when you look at real-world peptide compliance cases where audit failures traced back not to scientific misconduct, but to inadequate access controls and missing documentation. The portal’s mechanics are not just administrative tools. They are the evidence infrastructure that demonstrates study integrity.
Data privacy, regulatory requirements, and peptide procurement
While efficiency is crucial, data privacy and regulatory demands can complicate compliance. Understanding how top portals streamline these requirements alongside procurement is essential for researchers handling both sensitive participant data and proprietary compound sourcing.
The key regulatory frameworks that govern peptide research in the United States and internationally include:
- HIPAA (Health Insurance Portability and Accountability Act): Governs identifiable health data in studies involving human participants
- 21 CFR Part 11 (FDA Electronic Records Rule): Requires electronic signatures and audit trails to meet FDA standards for clinical and pre-clinical data
- GDPR (General Data Protection Regulation): Applies to EU participant data and increasingly affects US institutions with international collaborators
- IRB standards: Mandate informed consent documentation, protocol version tracking, and continuing review cycles
- FISMA: Federal Information Security Management Act requirements for federally funded research programs
For data privacy specifically, tools like REDCap serve as compliance portals for secure data capture, with features supporting HIPAA, 21 CFR Part 11, role-based permissions, logging, and data resolution workflows. REDCap’s adoption across academic medical centers is particularly notable: thousands of institutions globally have deployed it as their primary data compliance layer.
“REDCap’s combination of audit trail functionality, role-based access, and HIPAA-aligned data handling makes it one of the most widely trusted tools for research data management in the post-2020 regulatory environment.” This level of trusted infrastructure has measurable downstream effects on procurement integrity and data defensibility.
Pro Tip: Set up automated system notifications for access reviews, certification expirations, and protocol amendments. Many researchers assume their portal will flag issues proactively, but default notification settings in most systems are minimal. Customize alert thresholds for training expirations (30, 15, and 7 days out) and configure role-based alerts so PIs receive immediate notifications when a team member’s credentials lapse.
The connection between portal-based data privacy and research documentation is especially relevant in peptide procurement. When sourcing research-grade compounds, the documentation chain from certificate of analysis to study protocol to participant data must be seamless and auditable. A gap anywhere in that chain can trigger a regulatory finding.
Understanding peptide classifications also matters here, because different classes of peptides carry different regulatory documentation requirements. A research-use-only (RUO) peptide has a different compliance footprint than a compound under investigational new drug (IND) review. Your portal must be configured to track which category each compound falls into.
Consider a cautionary scenario: a mid-sized research institution conducts a peptide-based metabolic study. The team uses a general document management system rather than a purpose-built compliance portal. During an FDA audit, reviewers request timestamped records of every protocol amendment and the associated consent form versions. The institution cannot produce them in the required format. The study data is flagged as potentially unreliable. The entire dataset requires re-verification, and two years of work face potential disqualification. A properly configured compliance portal with version-controlled document storage and portal data security would have made that audit a straightforward, one-day event instead of a multi-month remediation effort.
Advanced features: Enterprise solutions for high-stakes peptide research
For larger institutions or those handling sensitive materials, generic solutions may fall short. Enterprise-grade portals are built to address the complexity of high-volume, multi-site, and high-stakes peptide research in ways that mid-tier platforms simply cannot match.
Advanced platforms like Cayuse and Dimensions provide enterprise compliance features including 21 CFR Part 11 adherence, risk assessment customization, foreign influence detection, and centralized evidence repositories. These capabilities are not incremental improvements. They represent a fundamentally different approach to compliance infrastructure.
| Feature | Mid-tier portal | Enterprise portal |
|---|---|---|
| Risk assessment tools | Basic checklist | Customizable, weighted scoring |
| Foreign influence detection | Not available | Automated disclosure flagging |
| Evidence repository | File upload | Centralized, searchable library |
| Dashboard customization | Template-based | Role-specific, configurable |
| Multi-site coordination | Limited | Full MOU and lead-site management |
| Regulatory update integration | Manual | Automated policy feed |
When should a research program consider upgrading to an enterprise platform? Several clear signals point toward that decision:
- Scale: More than 50 active protocols across multiple departments or sites
- Collaboration complexity: International partners, federal co-investigators, or industry sponsors
- Regulatory exposure: Studies involving IND applications, FDA device oversight, or controlled substances
- Integration requirements: Need to connect compliance data with electronic lab notebooks (ELNs), LIMS, or HR systems
- Audit history: Prior findings related to documentation gaps or access control failures
Research on AOD 9604 application in metabolic peptide studies illustrates exactly the kind of high-stakes scenario that benefits from enterprise-grade compliance infrastructure. Studies involving lipolytic peptides with potential clinical translation require airtight documentation from the early research phase forward, because any data weakness identified later can jeopardize IND submission.
The statistic worth noting: over 60% of research-intensive universities have either adopted or begun evaluating enterprise compliance platforms in the past five years, driven largely by increasing federal scrutiny of foreign influence in research and the growing complexity of multi-site clinical work.
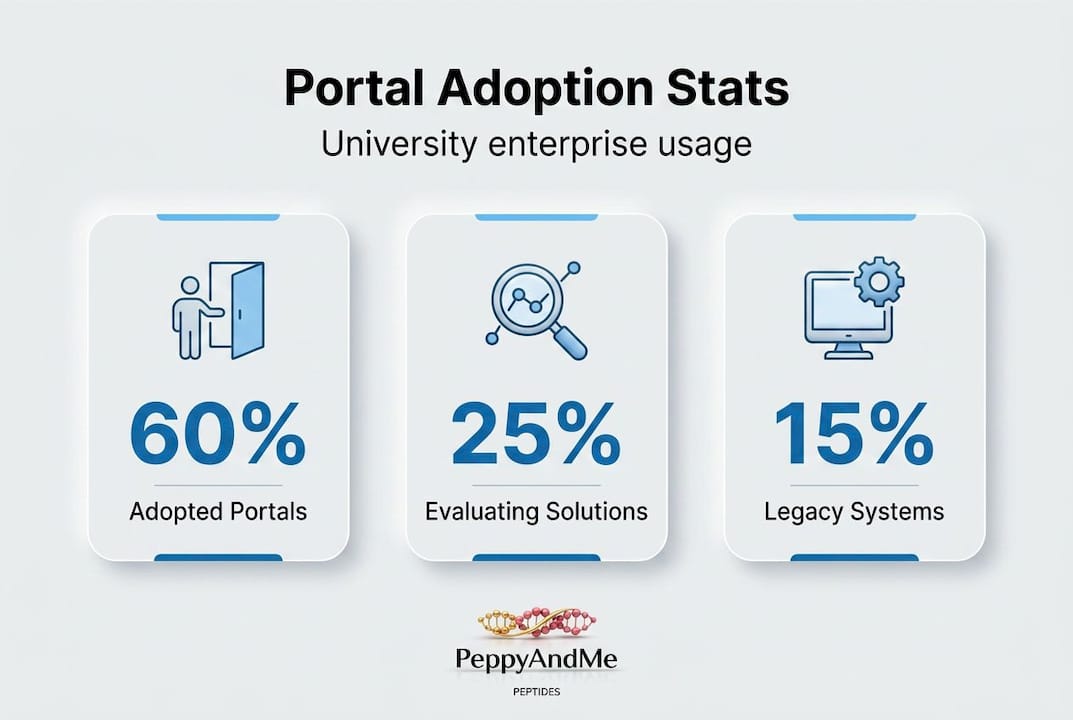
For advanced peptide therapy programs moving from exploratory to translational stages, an enterprise compliance portal is not an administrative upgrade. It is a scientific infrastructure decision that affects funding eligibility, publication credibility, and regulatory readiness.
Edge cases, challenges, and evolving compliance standards
Advanced solutions are not a guarantee against all compliance failures. Evolving regulations and institutional complexity present persistent new challenges, especially in the fast-moving regulatory environment surrounding peptide research.
The most commonly encountered edge cases in research compliance portal management include:
Multi-institution collaboration without proper agreements: When multiple institutions share a single protocol, lead institution designation and MOUs must be formally established. Non-primary institution collaborators may require explicit system recognition to access portal functions, and without it, workflow routing breaks down. External researchers working outside the primary portal environment may also fall outside the audit trail entirely.
Foreign influence disclosure gaps: Federal funding agencies now require detailed disclosure of international collaborations, foreign travel, and overseas funding sources. Most portals have added foreign influence modules in recent years, but institutions that customized their systems before these requirements emerged may have configuration gaps that create compliance exposure.
Dynamic consent and GDPR complexity: For studies involving EU participants or data, challenges persist in dynamic consent for GDPR research data, because standard IRB consent processes do not map cleanly onto GDPR’s right-to-withdraw and data portability requirements. Portals must be configured to manage consent versioning and participant withdrawal in ways that satisfy both US and EU frameworks simultaneously.
RUO vs. clinical peptide boundary management: Researchers working with peptides under RUO status sometimes apply data from those studies in ways that blur the line between preclinical exploration and clinical inference. Distinguishing RUO from clinical peptides to avoid regulatory pitfalls requires the portal to clearly tag compound status at the protocol level and prevent unapproved cross-referencing in publications or grant applications.
“The institutions that treat compliance portals as living systems, updated in real time as regulations shift, are the ones that avoid the gaps that cost other programs their funding and their data.” This perspective reflects what experienced compliance officers consistently report: static portal configurations are a liability in a dynamic regulatory environment.
Pro Tip: Subscribe to regulatory update feeds from FDA, NIH, and the EDPB (European Data Protection Board), and assign a specific team member to review and translate those updates into portal configuration changes at least quarterly. Do not rely on your portal vendor to push regulatory updates automatically. Many configuration decisions require institutional customization that vendors cannot make on your behalf.
Peptide compliance challenges are not going away as the field matures. If anything, the regulatory environment is becoming more rigorous, especially as more peptide compounds move from exploratory research into clinical investigation. The institutions that build compliance agility into their portal infrastructure now will be better positioned when the next regulatory shift arrives.
Our perspective: Compliance portals—asset or obstacle?
The resistance to compliance portal adoption is understandable. For research teams already managing demanding experimental work, the prospect of learning a new platform, reconfiguring workflows, and maintaining meticulous digital records can feel like a distraction from science. But that framing misses something important.
The institutions that have genuinely integrated compliance portals into their research culture, rather than treating them as a required nuisance, report a consistent secondary benefit: their studies attract more competitive funding. Federal agencies and private foundations increasingly use compliance infrastructure as a proxy for institutional readiness. A program that can demonstrate audit-ready documentation, clean access controls, and systematic protocol tracking signals maturity and reliability. That signal matters in grant review panels.
There is also a contrarian argument worth making directly. Researchers who delay portal adoption because they believe it will slow them down typically experience the opposite outcome. Manual compliance workflows introduce more errors, more revision cycles, and more last-minute protocol scrambles than any portal learning curve will. The time cost of adopting a portal is front-loaded and finite. The time cost of managing compliance without one compounds indefinitely.
Looking forward, the trajectory of compliance technology points toward AI-powered audit assistance, automated regulatory update integration, and direct linkage between compliance portals and procurement platforms. For peptide research specifically, the vision is a workflow where secure access for compliance wins means that a researcher can submit a protocol amendment, verify compound sourcing documentation, and confirm participant consent status from within a single integrated environment, with every action time-stamped, role-verified, and exportable.
Frame compliance as a competitive differentiator rather than an administrative tax, and the calculus changes entirely. Funding agencies notice. Collaborators notice. Journals conducting pre-publication data integrity checks notice. The research teams that treat compliance infrastructure as a strategic asset will find themselves with more collaboration opportunities, faster funding cycles, and more defensible data. That is not a coincidence. It is the system working as intended.
Optimize your peptide research workflow with trusted portals
Research compliance is only as strong as the procurement chain behind it. If your portal infrastructure is solid but your peptide sourcing introduces documentation gaps or quality uncertainty, the entire compliance architecture is undermined before your study begins.
Peppy&Me is built specifically for researchers who need both quality and accountability from their peptide supplier. Every compound is third-party tested for purity, mass accuracy, endotoxins, sterility, and heavy metals, with full lot and batch traceability from manufacturer to warehouse. That documentation integrates cleanly with your compliance portal records. For teams looking to scale their operations, the peptide brand partnership program offers a private-label pathway with the same backend rigor. And for researchers managing tight budgets without sacrificing quality, current peptide deals provide competitive access to premium-grade compounds. Compliance-ready sourcing and institutional-grade quality, without the premium markup.
Frequently asked questions
What is the main advantage of a research compliance portal in peptide studies?
It streamlines protocol submissions, tracks approvals, and centralizes evidence, improving transparency and reducing administrative delays. These portals automate compliance workflows including electronic submission, routing, progress tracking, and annual certifications, which is critical for maintaining study momentum.
How do compliance portals help with HIPAA and 21 CFR Part 11 requirements?
They feature role-based permissions, full logging, and support for secure electronic signatures, meeting major US data privacy laws. Tools like REDCap specifically support HIPAA and 21 CFR Part 11 compliance through structured access controls and complete audit logging.
What’s the difference between RUO and clinical peptides in compliance?
RUO peptides require less stringent processes, but clinical peptides must meet higher regulatory and data scrutiny. Compliance challenges persist in distinguishing RUO from clinical peptides, and portals help document and enforce that boundary at the protocol level.
Can compliance portals support multinational or collaborative research?
Yes, though multi-institution proceedings require lead institution designation and formal MOUs. External collaborators may also need explicit system recognition to participate fully in portal workflows without creating audit gaps.
What’s the biggest risk if compliance portals are not updated to new standards?
Outdated portals may expose projects to regulatory sanctions, security breaches, or disqualified data. Ongoing challenges in foreign influence and GDPR dynamic consent mean that static portal configurations become compliance liabilities as the regulatory environment continues to evolve.