Melanotan 10mg: Risks, Mechanisms, and Research Use

TL;DR:
- Melanotan 10mg is a cyclic peptide affecting multiple melanocortin receptors with safety concerns hindering its clinical use.
- Its receptor promiscuity complicates research, requiring rigorous sourcing and monitoring protocols.
- Alternatives like afamelanotide and bremelanotide offer more targeted, approved options for related studies.
Melanotan 10mg (melanotan II, or MT-II) occupies a uniquely contentious position in peptide research. It is neither a simple tanning agent nor a well-characterized therapeutic, yet its pharmacological complexity makes it one of the more instructive molecules in melanocortin biology. Development was halted due to a pattern of serious adverse events before Phase III trials could begin, including severe nausea in roughly 21% of subjects, spontaneous erections, and persistent pigmentation changes. For researchers in biochemistry, pharmacology, and aesthetics, understanding what melanotan 10mg is, what it does at the receptor level, and where the evidence actually stands is not optional. It is the minimum standard for responsible work with this compound.
Table of Contents
- What is melanotan 10mg? Structure and mechanism
- Regulatory landscape and clinical evidence
- Risks, adverse effects, and safety considerations
- Applications and experimental frameworks
- Controversies, gray-market realities, and research best practices
- The uncomfortable truth about melanotan 10mg in research
- Explore advanced research peptides and safety resources
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Distinct risks of melanotan II | Melanotan 10mg has unique side effect and safety challenges, including severe nausea and impact on mole monitoring. |
| No clinical approval | It remains unapproved for human or clinical use, with regulatory warnings and halted trials. |
| Photoprotection limitations | Cosmetic tanning effects do not equal the natural photoprotective benefits of endogenous melanin. |
| Role as research tool | It seeded the development of selective analogs like PT-141, offering both challenges and opportunities for research. |
| Ethical sourcing required | Proper validation and control procedures are essential when sourcing and using melanotan 10mg for research purposes. |
What is melanotan 10mg? Structure and mechanism
Melanotan II is a cyclic heptapeptide analog of alpha-melanocyte-stimulating hormone (alpha-MSH), a naturally occurring peptide derived from pro-opiomelanocortin (POMC). The synthetic modification that distinguishes MT-II from its parent molecule is cyclization, achieved via a lactam bridge between lysine and aspartic acid residues, which confers significantly greater receptor binding affinity and metabolic stability than the linear native hormone.
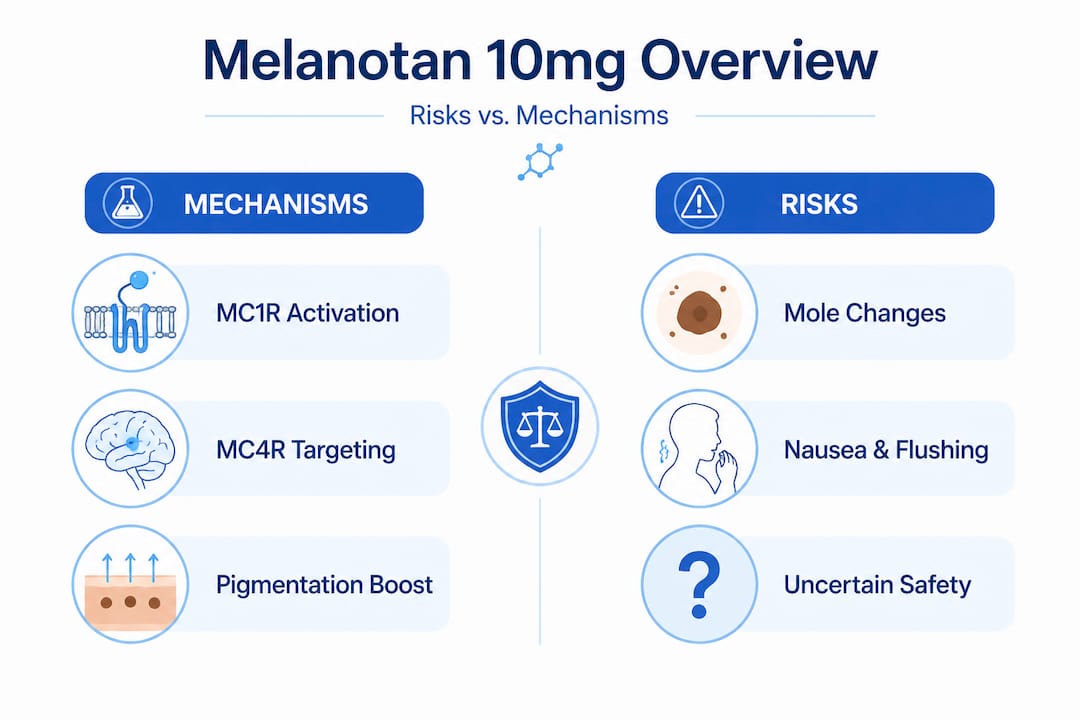
MT-II’s primary receptor targets are MC1R (melanocortin-1 receptor) and MC4R (melanocortin-4 receptor), though it also binds MC3R and MC5R with varying affinity. MC1R activation is responsible for the stimulation of melanogenesis in melanocytes, which drives the hyperpigmentation response researchers commonly associate with the compound. MC4R activation in the hypothalamus, however, underlies the sexual arousal effects, appetite suppression, and autonomic responses that define much of MT-II’s systemic risk profile.
Understanding this dual receptor engagement is essential. A researcher focusing solely on pigmentation outcomes may unintentionally expose their study model to significant MC4R-mediated effects, particularly in experimental designs using systemic routes of administration. The compound does not discriminate between these pathways, and neither can your protocol.
Afamelanotide (MT-I) offers a structurally and pharmacologically distinct comparison. It is a linear alpha-MSH analog with strong selectivity for MC1R, producing melanogenesis with minimal MC4R engagement. This receptor selectivity has enabled its regulatory approval: afamelanotide is FDA-approved for erythropoietic protoporphyria (EPP) under the brand name Scenesse. MT-I does not produce the sexual side effects, appetite changes, or autonomic disturbances associated with MT-II, which illustrates precisely why receptor selectivity matters in clinical and research translation.
| Feature | Melanotan II (MT-II) | Afamelanotide (MT-I) |
|---|---|---|
| Peptide structure | Cyclic heptapeptide | Linear 13-amino acid analog |
| Primary receptors | MC1R, MC4R, MC3R, MC5R | MC1R (selective) |
| Pigmentation effect | Yes | Yes |
| Sexual/autonomic effects | Yes (MC4R-mediated) | No |
| FDA approval | None | Yes (EPP/Scenesse) |
| EMA approval | None | Yes |
| Development status | Halted pre-Phase III | Approved |
| Common research dose | 0.5 to 1.0 mg/injection | Implant (16 mg subcutaneous) |
Key pharmacological characteristics of MT-II in research contexts include:
- High receptor promiscuity: Simultaneous MC1R/MC4R/MC3R agonism creates overlapping phenotypes that complicate endpoint isolation
- Cyclic structure advantage: Greater proteolytic resistance than linear analogs, resulting in longer biological activity windows
- Melanogenesis pathway: MC1R activation upregulates cAMP, stimulating tyrosinase and DOPA production in melanocytes
- Neuroendocrine engagement: MC4R activity in the hypothalamus modulates sexual function, feeding behavior, and cardiovascular tone
For researchers interested in peptides with cleaner mechanistic profiles, epitalon research peptide provides a useful reference for how targeted receptor engagement supports cleaner experimental design.
Regulatory landscape and clinical evidence
The clinical history of MT-II is short and instructive. Phase I and Phase II trials demonstrated that the compound produced consistent tanning and sexual arousal responses. However, the safety data generated in those trials was sufficient to prevent further progression. No Phase III trials were completed, and development as a pharmaceutical agent was abandoned. The FDA and EMA have both issued advisories against its use, and it currently holds no approved clinical indication in any jurisdiction.

The regulatory picture surrounding this compound is directly relevant to research operations:
| Compound | FDA Status | EMA Status | Approved Use | Warnings Issued |
|---|---|---|---|---|
| Melanotan II (MT-II) | Not approved | Not approved | None | Yes |
| Afamelanotide (MT-I) | Approved (2019) | Approved (2014) | EPP (Scenesse) | No |
| Bremelanotide (PT-141) | Approved (2019) | Not approved | HSDD (Vyleesi) | Specific populations |
This table clarifies the trajectory of melanocortin drug development. MT-II’s clinical failure was not the end of the pathway. Instead, it seeded the development of bremelanotide (PT-141), a metabolite of MT-II with more selective MC4R activity and a better-characterized safety profile. PT-141 received FDA approval in 2019 for hypoactive sexual desire disorder (HSDD) in premenopausal women, under the brand name Vyleesi. This regulatory history demonstrates that MT-II’s primary research value may lie less in direct application and more in serving as a parent scaffold for targeted analog development.
Regulatory context note: Researchers handling MT-II must recognize that working with an unapproved compound outside an institutional framework carries significant compliance obligations. Any adverse events observed in research models must be documented, and institutional review boards or ethical committees must be engaged before any human-adjacent research proceeds. FDA guidance for unapproved peptides under investigation is specific and non-negotiable.
For institutions operating under an investigational new drug (IND) framework or equivalent, the following steps represent the minimum compliance pathway for researchers working with unapproved peptides:
- Obtain institutional biosafety and ethics committee approval before initiating any dosing protocols
- Source material from a supplier with verifiable third-party testing, including endotoxin, sterility, and mass accuracy certificates
- Document the full chain of custody from manufacturer to lab, including lot and batch numbers
- Establish adverse event reporting procedures prior to any dosing, aligned with FDA or relevant national guidelines
- Maintain a research protocol that clearly defines endpoints, dosing ranges, and termination criteria
- Submit any human-relevant findings to appropriate regulatory or oversight bodies upon study completion
For context on how these compliance principles apply to other peptides in active research, the MOTS-c research guide and tesamorelin safe sourcing guidelines offer relevant parallels.
Risks, adverse effects, and safety considerations
The adverse effect profile of melanotan 10mg is one of the primary reasons its clinical development was terminated. Researchers must treat this profile not as a footnote but as a central variable in any experimental design involving the compound.
The most consistently reported effects across Phase I and II trials included:
- Nausea: Reported in the majority of subjects; severe nausea in 21% of subjects, representing a clinically significant incidence rate that contributed directly to the development halt
- Flushing and facial erythema: Common, typically onset within 30 to 60 minutes of dosing
- Fatigue: Reported broadly; mechanism likely involves MC3R and MC4R activity in central nervous system circuits
- Spontaneous erections: A direct MC4R-mediated effect; clinically described as priapism in edge cases
- Hyperpigmentation: Expected pharmacodynamic response, but includes darkening and growth of pre-existing moles and nevi, which creates a serious confounding issue for melanoma screening in long-term research subjects
- Oral mucosal pigmentation: Documented case reports describe persistent oral mucosa pigmentation lasting more than three months following cumulative dosing of approximately 12.8 mg, even after cessation
The mole darkening and growth concern deserves focused attention in any research design. When MT-II stimulates melanocyte activity non-selectively, it does not distinguish between normal melanocytes and atypical ones. The result is that abnormal nevi may darken or enlarge in ways that visually mimic benign tanning responses, masking potential melanoma signals. This is not a theoretical risk. It represents a tangible gap in monitoring capability that any responsible research protocol must address.
Pro Tip: Before initiating any study involving MT-II, establish a full baseline dermoscopy and mole-mapping session for all relevant research subjects. Schedule repeat assessments at each major study interval. Document every pigmented lesion with standardized photography and dermoscopic data so that any change can be attributed to study compound rather than occult pathology. This step is not optional for rigorous research.
The distinction between rodent and human pharmacokinetic data is also worth stating clearly. Much of the foundational literature on MT-II was developed in murine models, where receptor expression patterns and metabolic rates differ meaningfully from human physiology. Translating rodent dosing benchmarks directly to human-relevant protocols is a common and significant methodological error. Researchers should treat rodent data as hypothesis-generating and prioritize available human Phase I/II safety data when designing monitoring thresholds.
For ongoing research developments, adverse event summaries, and updated safety literature, the lab insights blog provides regularly updated resources aligned with current research practice.
Applications and experimental frameworks
Despite its regulatory status, melanotan 10mg retains value as a research tool precisely because of its pharmacological complexity. Its ability to simultaneously activate multiple melanocortin receptor subtypes makes it a useful probe compound for studying the intersection of pigmentation biology, neuroendocrine regulation, and metabolic function.
Empirical tanning benchmarks from available literature provide some research-usable reference points:
- Visible tanning appears within 1 to 2 weeks of standard research dosing protocols when combined with UV exposure
- Peak tanning response is typically observed at 4 to 6 weeks of continued dosing
- MT-II synergizes with UV radiation to accelerate and intensify melanogenesis, but it does not provide photoprotection equivalent to naturally increased melanin; the increase in melanin density from MT-II stimulation does not replicate the full ultrastructural distribution of UV-adaptive melanin
This last point carries significant implications for any aesthetic or dermatological research framing MT-II as a skin-protective agent. The claim of photoprotection has circulated widely outside the scientific literature, but the evidence does not support it at the level required for any valid research conclusion.
Key areas where MT-II provides legitimate experimental value include:
- Receptor subtype dissection: Using MT-II alongside selective MC1R and MC4R antagonists allows researchers to delineate which effects are subtype-specific, providing mechanistic data that selective agonists alone cannot generate
- Melanogenesis kinetics: Dose-response studies examining the time course of tyrosinase activation and DOPA production remain an active area, particularly in skin biology research
- Neuroendocrine interaction studies: MC4R engagement by MT-II provides a pharmacological handle on appetite regulation, energy homeostasis, and sexual function circuits that are of interest in metabolic disease research
- Analog benchmarking: MT-II serves as a reference compound against which more selective analogs (e.g., PT-141, afamelanotide) can be characterized in head-to-head frameworks
Pro Tip: Design protocols that explicitly separate cosmetic melanogenic phenotypes from systemic MC-related phenotypes. Using MC4R-selective antagonists as co-treatments in separate experimental arms allows you to attribute systemic effects to MC4R while isolating the MC1R-driven pigmentation data. This approach dramatically increases the interpretive value of your findings, particularly when characterizing a new analog against MT-II as a reference.
The AOD-9604 research guide offers a useful parallel for how researchers approach controlled experimental design with metabolically active peptides.
Controversies, gray-market realities, and research best practices
The scientific controversies surrounding melanotan 10mg are well-documented in peer-reviewed literature. The debate is not simply about whether it works. It concerns whether the available evidence justifies any continued research application, particularly given the volume of gray-market product circulating under its name.
“Contrasting views from the literature and practice community reflect the complexity of MT-II’s status: it is recognized as a potent research tool that seeded the development of PT-141, but unregulated products carry risks of contamination, dosing inconsistency, and unknown long-term safety, while the relationship between MT-II use and melanoma causality remains debated even as screening reliability is demonstrably impaired.”
The gray-market reality is straightforward. Melanotan 10mg is widely available through non-regulated channels, frequently mislabeled, and often produced without any quality controls. Studies on off-market peptide products have identified issues including incorrect molecular weights, bacterial contamination, heavy metal presence, and significant dosing inconsistencies between labeled and actual content. These issues do not merely affect research reproducibility. They create genuine harm potential for anyone involved in their use.
Actionable best practices for researchers working with MT-II in a controlled environment:
- Validate every product batch using third-party mass spectrometry and HPLC analysis before any experimental use; do not rely solely on supplier-provided certificates of analysis
- Confirm endotoxin levels using Limulus Amebocyte Lysate (LAL) testing to rule out pyrogen contamination, which produces confounding inflammatory responses
- Maintain full chain-of-custody documentation from order placement through receipt, storage, and use, with lot and batch numbers logged at every stage
- Report adverse events to appropriate oversight bodies in real time; do not wait until study completion to disclose unexpected findings
- Adhere to FDA guidance on unapproved investigational substances; consult institutional legal and compliance resources before initiating any novel protocol
- Schedule routine impurity testing at multiple study intervals to detect product degradation, which is especially relevant given MT-II’s sensitivity to temperature and light exposure
For further guidance on sourcing quality research peptides and avoiding the pitfalls of gray-market supply chains, peptide sourcing best practices provides a detailed sourcing framework directly applicable to MT-II procurement decisions.
The argument that MT-II retains unique value as a research tool is strongest when the compound is handled with this level of rigor. Without it, the data generated is not just unreliable. It is potentially dangerous.
The uncomfortable truth about melanotan 10mg in research
The prevailing narrative around melanotan 10mg tends toward one of two extremes. Either it is dismissed as a poorly understood gray-market tanning peptide with no legitimate research place, or it is championed as an underappreciated tool being suppressed by overcautious regulators. Neither position reflects the actual state of the evidence.
The uncomfortable reality is that MT-II is both genuinely pharmacologically interesting and genuinely poorly characterized in ways that matter for research integrity. Its receptor promiscuity, which makes it attractive as a probe compound, is also precisely what makes its safety profile so difficult to manage in practice. You cannot activate MC1R without engaging MC4R at any therapeutically relevant dose, and that constraint alone limits the interpretive cleanliness of most experimental designs built around it.
What the literature and current research peptide frameworks consistently show is that the compounds derived from MT-II, particularly bremelanotide/PT-141 and afamelanotide, offer more tractable research pathways for most objectives. If you are studying pigmentation biology, afamelanotide’s MC1R selectivity and approved clinical status provide a cleaner experimental substrate with better-characterized safety boundaries. If you are studying sexual function or MC4R-mediated neuroendocrinology, bremelanotide provides a more selective tool with actual Phase III data and FDA approval behind it.
This does not mean MT-II has no place in 2026 research. It means the threshold for justifying its use over more selective, better-characterized analogs should be high. Labs that choose to work with it should have advanced dermoscopic monitoring in place, institutional ethical oversight, validated sourcing with full third-party testing, and protocols specifically designed to account for multi-receptor engagement. Most research questions that appear to require MT-II can be answered more cleanly with its descendants. The ones that genuinely require MT-II as a reference or probe compound are worth pursuing with full rigor, not as a shortcut.
Critical skepticism is not the same as blanket prohibition. It is the standard that separates high-quality research from noise in the literature.
Explore advanced research peptides and safety resources
Researchers who have worked through the pharmacological and regulatory complexities of melanotan 10mg know that quality sourcing and access to rigorous scientific resources are not secondary concerns. They are fundamental to producing defensible data.
Peppy&Me’s research peptide catalog provides verified access to research-grade peptides with full third-party testing documentation, traceable lot numbers, and same-day shipping for orders placed before 2 PM. For institutions or entrepreneurs interested in building their own branded research supply infrastructure, the private label peptides partnership program offers backend fulfillment and operational support. Researchers handling sensitive compound data and procurement records will also find practical guidance through the peptide security guide, which covers data protection best practices for research procurement operations.
Frequently asked questions
How long do visible tanning effects of melanotan 10mg last in studies?
Tanning effects typically become visible within 1 to 2 weeks of research dosing and reach peak intensity around 4 to 6 weeks, with UV co-exposure accelerating this timeline.
Does melanotan 10mg provide photoprotection equivalent to natural melanin?
No. Research confirms that melanotan 10mg does not deliver the same photoprotection as natural melanin; MT-II-induced pigmentation lacks the full ultrastructural distribution of UV-adaptive melanin that underlies genuine photoprotection.
What major safety risks should researchers watch when studying melanotan 10mg?
Key risks include severe nausea in 21% of subjects, flushing, fatigue, spontaneous erections, priapism in edge cases, and mole darkening or growth that can impair melanoma screening reliability throughout and after the study period.
Is melanotan 10mg approved for any clinical use?
No. There are no FDA or EMA approvals for any clinical indication; development was halted before Phase III trials, and regulatory warnings have been issued against its unsupervised use in any jurisdiction.