MOTS-c 10mg: Guide to Benefits, Mechanism, and Research Use

TL;DR:
- MOTS-c is a mitochondrial microprotein that mimics exercise by activating AMPK pathways.
- A 10mg daily IV dose improved insulin sensitivity in a small human trial.
- Safety, long-term effects, and optimal dosing require further research before widespread use.
MOTS-c is not a conventional peptide. It originates from mitochondrial DNA, not the nuclear genome, making it a rare class of signaling molecule called a mitochondrial microprotein. What makes it genuinely remarkable is that MOTS-c acts as an exercise mimetic, triggering metabolic adaptations typically associated with physical activity without requiring mechanical stress on the body. For researchers studying metabolic disease and for biohackers pushing the boundaries of performance science, the 10mg dosing protocol has become a key reference point. This guide breaks down the science, the evidence, and the responsible application of MOTS-c at that dose.
Table of Contents
- What is MOTS-c and why the 10mg dose matters
- MOTS-c mechanisms of action: AMPK and beyond
- What MOTS-c 10mg does: Evidence from animal and human studies
- Limitations, safety, and practical considerations for research and self-experimentation
- Why MOTS-c 10mg protocols need better data: A research and biohacker reality check
- Secure reliable MOTS-c for your research
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| MOTS-c is an exercise mimetic | It activates AMPK pathways like exercise but without physical stress. |
| 10mg dose has preliminary support | A 10mg daily dose improved metabolic parameters in a small human trial. |
| Animal studies show broad benefits | MOTS-c enhanced weight control, protected muscle, and reduced inflammation in preclinical models. |
| Clinical data are limited | Widespread human use is not yet supported by robust clinical trials. |
| Responsible research is vital | Ensure thorough safety and efficacy evaluation before self-experimenting or implementing research protocols. |
What is MOTS-c and why the 10mg dose matters
MOTS-c, short for Mitochondrial Open Reading Frame of the 12S rRNA-c, was first identified and characterized in a landmark 2015 paper published in Cell Metabolism by Changhan David Lee and colleagues. Unlike most peptides, it is encoded within the mitochondrial genome, specifically within the 12S ribosomal RNA gene. This origin alone sets it apart from nuclear-encoded hormones and peptides, and it explains why MOTS-c is considered a retrograde signaling molecule, one that communicates from the mitochondria back to the nucleus to regulate gene expression and cellular energy balance.
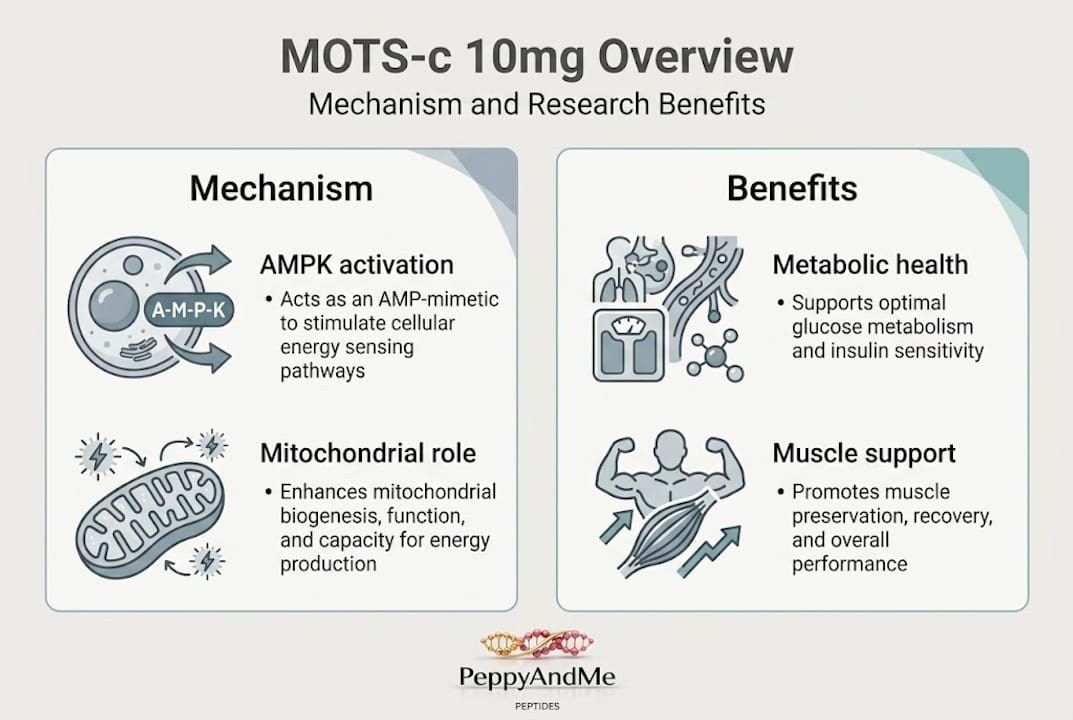
At its core, MOTS-c activates AMP-activated protein kinase (AMPK), the master regulator of cellular energy homeostasis. AMPK activation promotes glucose uptake, fatty acid oxidation, and mitochondrial biogenesis. These are the same pathways that physical exercise activates. The difference is that MOTS-c does this at the molecular level without requiring the mechanical and cardiovascular demands of exercise itself, which makes it a powerful research tool for isolating metabolic effects from physical performance variables.
Key properties of MOTS-c at a glance:
- Origin: Encoded in mitochondrial DNA, 12S rRNA gene
- Classification: Mitochondrial-derived peptide (MDP), microprotein
- Primary pathway: AMPK activation, mitonuclear signaling
- Endogenous relevance: Circulating levels decline with age, obesity, and type 2 diabetes (T2D)
- Research focus: Metabolic regulation, insulin sensitivity, muscle preservation, aging
The 10mg dose became the reference point because of a small proof-of-concept trial in which 10 overweight men received 10mg/day intravenously for seven days, resulting in measurably improved insulin sensitivity and glucose clearance. While this is a very small sample, it is currently the most direct human data available and forms the basis for most research-oriented and biohacker protocols. Conditions associated with reduced endogenous MOTS-c levels include T2D, obesity, and age-related metabolic decline, which is precisely why researchers are interested in exogenous supplementation as a potential intervention strategy. Explore more peptide research insights to see how MOTS-c fits within the broader landscape of mitochondrial peptide science.
Statistic callout: In the only published human pilot trial, 10mg/day IV MOTS-c for 7 days improved insulin sensitivity and glucose clearance in overweight men, with no serious adverse events reported during the study period.
MOTS-c mechanisms of action: AMPK and beyond
Understanding how MOTS-c works at the cellular level is essential for both rigorous research design and informed self-experimentation. The mechanism is more nuanced than simple AMPK agonism, and that nuance matters when comparing MOTS-c to existing metabolic drugs.
MOTS-c activates AMPK primarily through the generation of endogenous AICAR (5-aminoimidazole-4-carboxamide ribonucleotide), an intermediate in the folate cycle within mitochondria. This is a key distinction: AMPK activation via endogenous AICAR is considered more physiologically relevant than direct AICAR supplementation or metformin, both of which activate AMPK through different and less targeted mechanisms. The endogenous pathway MOTS-c uses mirrors what happens naturally during exercise, which is why its exercise-mimetic label is scientifically defensible rather than marketing language.
| Mechanism | MOTS-c | Exogenous AICAR | Metformin |
|---|---|---|---|
| AMPK activation route | Endogenous AICAR via folate cycle | Direct AICAR supplementation | Complex I inhibition |
| Physiological relevance | High (mirrors exercise signaling) | Moderate | Lower |
| Nuclear signaling | Yes (mitonuclear retrograde) | Limited | No |
| Fatty acid respiration effect | Neutral in some diabetic models | Increases | Variable |
| Senotherapeutic potential | Emerging evidence | Not established | Limited |
Beyond AMPK, MOTS-c translocates to the nucleus under cellular stress conditions, where it directly regulates gene expression. This nuclear role enables what researchers call retrograde mitonuclear signaling, a feedback loop in which mitochondria communicate their functional status to the nucleus to adjust transcription accordingly. This positions MOTS-c as more than a simple metabolic activator; it is a regulatory signal that integrates mitochondrial health with nuclear gene programs.
One important edge case worth noting: MOTS-c does not affect fatty acid-supported mitochondrial respiration or reactive oxygen species (ROS) production in certain diabetic models. This means its metabolic effects are not uniformly broad. Researchers should not assume MOTS-c will improve all aspects of mitochondrial function across all disease contexts.
“MOTS-c activates AMPK through endogenous AICAR generation, a mechanism that more closely mirrors physiological exercise signaling than either direct AICAR or metformin administration, while also enabling retrograde mitonuclear communication that neither drug replicates.”
Pro Tip: When designing MOTS-c experiments, consider that its AMPK activation route is substrate-dependent and context-sensitive. Running parallel arms with direct AICAR or metformin controls will help isolate MOTS-c-specific effects and strengthen your data interpretation. Learn more about AMPK activation explained through the Peppy&Me research library.
What MOTS-c 10mg does: Evidence from animal and human studies
With the molecular mechanisms outlined, understanding real-world biological effects at the 10mg dose is the logical next step. The evidence base currently spans rodent models and one small human pilot, and both offer meaningful but distinct insights.
In a T2D rat model, MOTS-c treatment reduced weight gain, improved glucose handling, reversed left ventricular (LV) hypertrophy, and restored carbohydrate-supported mitochondrial respiration and citrate synthase activity in heart mitochondria. These are significant findings because LV hypertrophy is a serious cardiac complication of T2D, and restoring citrate synthase activity indicates genuine improvement in mitochondrial oxidative capacity, not just surface-level metabolic markers.

In a separate immobilization mouse model, MOTS-c at 7.5 mg/kg twice daily reduced muscle mass loss from approximately 15% down to approximately 5%, normalized p-AKT/FOXO signaling, reduced inflammatory cytokines including IL-1β and IL-6, and inhibited fat infiltration into muscle tissue via the PPAR pathway. For researchers studying muscle atrophy, disuse-related deconditioning, or sarcopenia, these results are particularly compelling.
| Model system | Dose used | Key outcomes |
|---|---|---|
| T2D rat model | Not specified per kg | Reduced weight gain, improved glucose, reversed LV hypertrophy, restored mitochondrial respiration |
| Immobilization mouse model | 7.5 mg/kg 2x/day | Attenuated muscle loss, reduced IL-1β/IL-6, inhibited fat infiltration |
| Human pilot (overweight men) | 10mg/day IV x7 days | Improved insulin sensitivity, improved glucose clearance |
What the current evidence supports vs. what remains unproven:
- Well-established in preclinical models: AMPK activation, improved glucose metabolism, muscle mass preservation, anti-inflammatory effects, and cardiac protection in T2D contexts.
- Supported but limited in humans: Improved insulin sensitivity and glucose clearance at 10mg/day IV over 7 days in a small cohort.
- Not yet established: Long-term safety in humans, optimal dosing frequency, subcutaneous vs. intravenous bioavailability equivalence, and effects in healthy non-diabetic populations.
- Unproven in humans: Muscle preservation benefits, cardiac protection, anti-aging effects, and performance enhancement, despite strong animal data.
- Unknown: Chronic use effects, dose-response relationship in humans, and interaction with existing metabolic medications.
Explore animal study summaries on the Peppy&Me blog to contextualize these findings within the broader peptide research literature.
Limitations, safety, and practical considerations for research and self-experimentation
Knowing the benefits is only responsible if limitations and caution are also made clear. The MOTS-c research landscape is genuinely exciting, but the gap between preclinical promise and clinical validation is wide, and both researchers and biohackers need to navigate it carefully.
The most fundamental limitation is straightforward: no large-scale human clinical trials exist. The entire human evidence base rests on a single proof-of-concept study with 10 participants. That is not enough to establish efficacy, determine optimal dosing, or characterize safety in diverse populations. The preclinical consensus is strong, anchored by the foundational Lee and Cohen 2015 Cell Metabolism paper, but animal-to-human translation for metabolic peptides is notoriously unreliable. Many compounds that perform brilliantly in rodent models fail to replicate those results in human trials.
What we do not yet know about MOTS-c:
- Safe upper dose limits in humans
- Effects of chronic administration beyond 7 days
- Subcutaneous bioavailability compared to intravenous delivery
- Interaction effects with insulin, metformin, or GLP-1 agonists
- Tissue-specific distribution and clearance rates in humans
- Long-term impact on endogenous MOTS-c production
The biohacker community has moved faster than the science, with anecdotal reports of higher doses and longer cycles circulating widely online. These protocols lack randomized controlled trial (RCT) backing and should not be treated as validated. Extrapolating rodent doses directly to humans without allometric scaling is a common error that can lead to meaningfully different exposure levels than intended.
For researchers, the ethical and regulatory framework is equally important. MOTS-c is not approved for therapeutic use in any jurisdiction. Institutional review board (IRB) approval, proper informed consent, and adherence to Good Laboratory Practice (GLP) standards are non-negotiable for any human-facing research.
Pro Tip: When designing a MOTS-c protocol, start with the lowest dose that produced measurable effects in the published literature and build in washout periods. Document all observations systematically, including any subjective reports in self-experimentation contexts, and cross-reference with the limitations and safety resources available through Peppy&Me’s research library before finalizing any protocol.
For self-experimenters specifically, the intravenous route used in the human pilot is not practical or safe outside of clinical settings. Subcutaneous administration is the common alternative in biohacker circles, but direct bioavailability comparisons between IV and SC routes for MOTS-c have not been published. That is a significant unknown that should factor into any personal protocol design.
Why MOTS-c 10mg protocols need better data: A research and biohacker reality check
There is a pattern in peptide research that repeats itself: a mechanistically compelling compound emerges from preclinical work, a small human study generates encouraging signals, and enthusiasm rapidly outpaces the evidence. MOTS-c is following that pattern precisely. The biology is genuinely interesting, the animal data is robust, and the 10mg human pilot is encouraging. But it would be a mistake to treat those findings as proof of concept for widespread use.
The clinical translation lag in peptide science is real and persistent. Rodent metabolism differs from human metabolism in ways that consistently undermine direct dose extrapolation and effect size predictions. The cardiac and muscle preservation data from animal models are particularly susceptible to this problem because rodent cardiac physiology and muscle fiber composition differ substantially from human equivalents.
What concerns us most is the conflation of mechanism with outcome in biohacker communities. Knowing that MOTS-c activates AMPK does not mean a given protocol will produce the metabolic benefits seen in controlled studies. Dose, route, timing, individual metabolic status, and baseline MOTS-c levels all modulate the response in ways that are not yet characterized. Reviewing critical appraisal of peptide research is a good starting point for developing a more rigorous framework.
The responsible position is this: MOTS-c 10mg protocols deserve continued investigation through properly powered RCTs. Until that data exists, both researchers and self-experimenters should treat current protocols as exploratory, document everything, and resist the urge to generalize small-sample findings into confident recommendations.
Secure reliable MOTS-c for your research
Responsible MOTS-c research starts with sourcing peptides you can actually trust. At Peppy&Me, every batch of research-grade MOTS-c is third-party tested for purity, mass accuracy, endotoxins, sterility, and heavy metals, with full lot and batch traceability from manufacturer to warehouse.
Whether you are designing a metabolic study or refining a personal protocol, precision matters. Use the dose calculator to ensure accurate preparation every time, and explore the research library for protocol guidance and peptide handling best practices. For labs or entrepreneurs looking to scale, the private label peptide sourcing program offers a fully supported backend for building your own research peptide brand. Same-day shipping for orders placed before 2 PM keeps your research timeline on track.
Frequently asked questions
What is the optimal MOTS-c dose for humans based on available studies?
The only published human data used 10mg/day IV for 7 days, but this single small trial is insufficient to define an optimal dose, and no dose-response studies in humans have been published.
Are there any large clinical trials of MOTS-c 10mg?
No large-scale clinical trials of MOTS-c exist; current human evidence is limited to a single proof-of-concept study in 10 overweight men and observational data showing reduced endogenous levels in metabolic disease.
What benefits has MOTS-c shown in preclinical models?
In a T2D rat model, MOTS-c reduced weight gain and improved glucose handling, while in an immobilization mouse model, it attenuated muscle mass loss from approximately 15% to 5% and reduced inflammatory cytokines including IL-1β and IL-6.
Is MOTS-c safe to use for self-experimentation?
Safety is not established for MOTS-c in humans; no large-scale trials have characterized its risk profile, chronic use effects are unknown, and intravenous administration outside clinical settings carries additional procedural risks.