AOD 9604 generated real excitement when early animal studies reported dramatic reductions in body weight and restored metabolic function in obese rodent models. Then human trials arrived, and the results were far more modest. That gap between preclinical promise and clinical performance has left many researchers uncertain about how to interpret the literature, design rigorous protocols, and source verified material. This guide cuts through the noise. It covers AOD 9604’s biochemical structure, what the animal and human data actually show, how to approach the 2mg research standard, and the critical caveats every investigator needs to understand before moving forward.
Table of Contents
- Understanding AOD 9604: Structure, mechanism, and formulation
- Preclinical evidence: What animal studies reveal about AOD 9604
- Human clinical trials: Weight loss outcomes, dose response, and safety data
- Practical application in research: Acquisition, protocol design, and critical caveats
- Expert perspective: Why AOD 9604 is more than preclinical hype (and what researchers really need to know)
- Ready to advance your AOD 9604 research?
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Structure and mechanism | AOD 9604 is a stabilized analog of GH Fragment 176-191 with specific modifications for increased research utility. |
| Robust preclinical data | Animal studies show dramatic weight loss and metabolic effects, but translation to humans is limited. |
| Human efficacy and safety | Clinical trials indicated modest, non-linear effects on weight with an excellent safety profile. |
| Practical research guidance | Sourcing, dose setting, and protocol design require careful consideration due to regulatory and translational nuances. |
| Expert perspective | AOD 9604 exemplifies why critical appraisal and documentation are vital in peptide research. |
Understanding AOD 9604: Structure, mechanism, and formulation
With the challenges clarified, let’s first establish what AOD 9604 actually is and why its formulation and mechanism matter for your experiments.
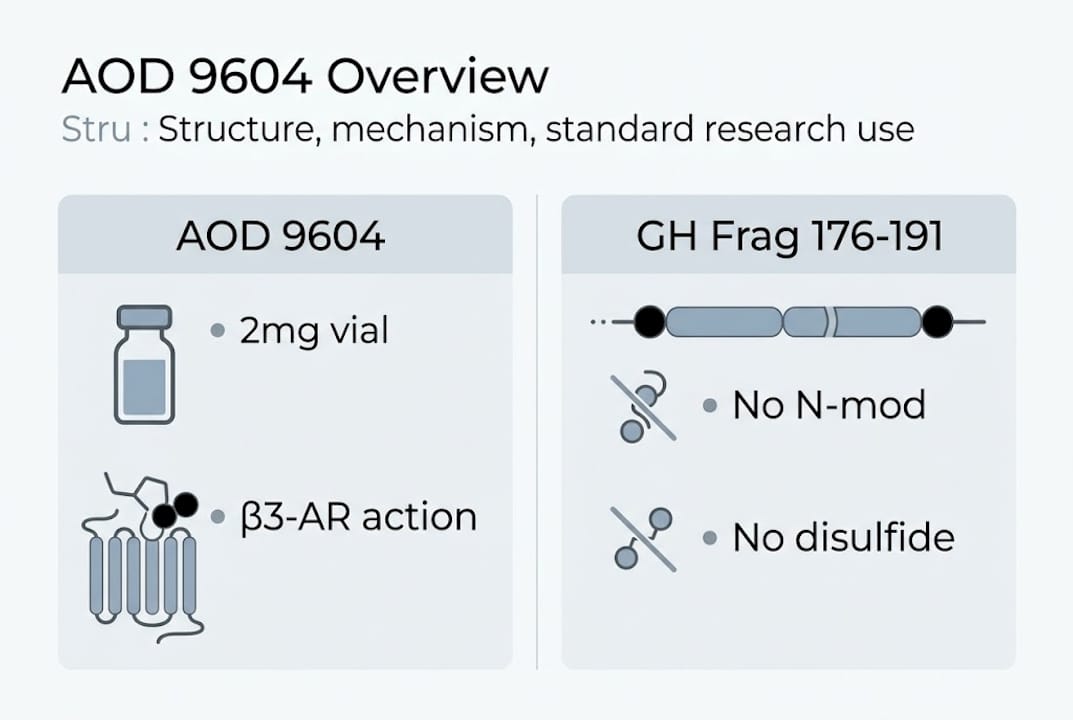
AOD 9604 is a synthetic peptide fragment (Tyr-hGH 177-191) of human growth hormone’s C-terminal lipolytic domain, carrying the CAS number 221231-10-3 and a molecular weight of approximately 1815 Da. The sequence spans residues 177 to 191 of hGH, with a tyrosine residue added at the N-terminus for improved stability and pharmacological performance. A disulfide bridge between Cys182 and Cys189 maintains the peptide’s three-dimensional conformation, which is critical for receptor binding.
Compared to the parent GH Fragment 176-191, AOD 9604 offers two structural improvements. The tyrosine addition increases metabolic stability against proteolytic degradation. The preserved disulfide bridge ensures the active conformation is maintained under physiological conditions. Both modifications contribute to more reproducible experimental outcomes when working with in vitro or in vivo models.
Key structural features of AOD 9604:
- Sequence: Tyr-hGH 177-191 (15 amino acids)
- Molecular weight: ~1815 Da
- Disulfide bridge: Cys182-Cys189
- CAS number: 221231-10-3
- Mechanism target: β3-adrenergic receptor (β3-AR)
The mechanism of action centers on β3-AR activation. Acutely, AOD 9604 stimulates lipolysis by activating β3-AR in adipose tissue, promoting fatty acid release. Chronically, it appears to downregulate lipogenic enzymes and restore β3-AR sensitivity in obese models where receptor expression is typically blunted. Crucially, AOD 9604 does not activate the GH receptor, meaning it avoids IGF-1 elevation and insulin resistance, which are common concerns with full-length GH analogs.
The 2mg vial is the standard research unit because it aligns with doses used in preclinical and early clinical protocols, allows precise reconstitution for multiple experimental runs, and minimizes waste when working with small cohorts or in vitro assays.
| Feature | GH Fragment 176-191 | AOD 9604 |
|---|---|---|
| N-terminal modification | None | Tyrosine added |
| Disulfide bridge | Present | Present |
| Proteolytic stability | Moderate | Improved |
| β3-AR activity | Yes | Yes |
| GH receptor activation | No | No |
Pro Tip: Before reconstituting your 2mg vial, verify peptide integrity using HPLC purity data and mass spectrometry confirmation from your supplier. Sequence errors or oxidized disulfide bridges will compromise your results and make data interpretation unreliable.
Preclinical evidence: What animal studies reveal about AOD 9604
With the structure and mechanism clear, it’s vital to assess what animal studies actually demonstrate about AOD 9604’s research value.
Rodent studies produced some of the most striking metabolic data seen in peptide research. Obese Zucker rats receiving oral 500 mcg/kg/day showed a 50% reduction in weight gain compared to untreated controls, a result that drove significant early interest. In diet-induced obese mice, subcutaneous administration restored β3-AR expression in adipose tissue and increased lipolytic activity, suggesting a receptor-level correction rather than simple caloric suppression.
The β3-AR dependency was confirmed through knockout studies. Chronic AOD 9604 reduces weight and lipolysis in wild-type obese mice but produces no effect in β3-AR knockout mice, which directly confirms the receptor-dependent mechanism and rules out off-target explanations.
“The β3-AR knockout data is arguably the most important finding in AOD 9604 preclinical research. It tells you exactly what the peptide needs to work, and it tells you why human translation is inherently complicated.”
The problem is species-specific receptor distribution. Rodents express β3-AR abundantly in brown adipose tissue, which is metabolically active and highly responsive. Humans have far less brown adipose tissue and lower β3-AR density in white fat depots. This fundamental anatomical difference is why rodent efficacy data cannot be directly extrapolated to human outcomes.
Critical design considerations for replicating preclinical results:
- Match the obesity model carefully. Diet-induced obesity and genetic obesity (Zucker rats) show different receptor profiles.
- Control for baseline β3-AR expression in your animal cohort before treatment begins.
- Use species-appropriate dose scaling rather than direct mg/kg conversion from rodent to human.
- Measure both acute lipolysis markers and chronic receptor expression changes as separate endpoints.
- Include β3-AR knockout controls if your goal is mechanistic confirmation rather than efficacy screening.
- Document housing conditions, diet composition, and activity levels, all of which affect adipose receptor density.
| Model | Route | Dose | Key Outcome |
|—|—|—|
|—|
| Obese Zucker rat | Oral | 500 mcg/kg/day | 50% weight gain reduction |
| Diet-induced obese mouse | Subcutaneous | Variable | Restored β3-AR, increased lipolysis |
| β3-AR knockout mouse | Subcutaneous | Variable | No effect confirmed |
Human clinical trials: Weight loss outcomes, dose response, and safety data
Animal studies set high expectations, but human data is essential for translational guidance.
Six randomized controlled trials involving approximately 900 subjects were conducted across the AOD 9604 clinical program. Phase IIa enrolled 300 obese participants over 12 weeks using oral dosing at 1 mg/day. The result was a statistically significant but modest 2.8 kg weight loss compared to 0.8 kg in the placebo group. That’s a real signal, but it’s a long way from the 50% weight gain reduction seen in rodents.

Phase IIb, a larger placebo-controlled trial, failed to meet its primary endpoint. The development program was discontinued. This is the outcome that most peptide research summaries underreport, and it matters enormously for how you frame your own research questions.
Key takeaways from the clinical trial program:
- Non-linear dose response: higher doses did not produce proportionally greater weight loss
- Oral bioavailability was used in trials, while most current research protocols use subcutaneous delivery
- Trial duration (12 weeks) may have been insufficient to detect longer-term metabolic adaptations
- Subject heterogeneity in β3-AR expression was not controlled for in published designs
- Endpoint selection focused on body weight rather than adipose-specific lipolysis markers
The established safety data across these trials is genuinely robust. No IGF-1 elevation, glucose intolerance, insulin resistance, or GH-like effects were observed at any tested dose. AOD 9604 was well tolerated across all six trials, which is a meaningful finding for researchers designing safety-focused studies.
| Trial Phase | N | Duration | Outcome |
|---|---|---|---|
| Phase IIa | 300 | 12 weeks | 2.8 kg vs 0.8 kg placebo (significant) |
| Phase IIb | ~600 | 24 weeks | Failed primary endpoint |
| Safety profile | ~900 | Pooled | No GH-like adverse events |
Statistic callout: Across all six RCTs, zero cases of IGF-1 elevation or insulin resistance were recorded, making AOD 9604 one of the best-characterized peptides for metabolic safety in the clinical literature.
The 2mg dose used in research kits reflects the experimental range, not a therapeutic standard. Researchers should treat it as a starting benchmark, not a validated clinical dose.
Practical application in research: Acquisition, protocol design, and critical caveats
Understanding the evidence enables more rigorous design of your own AOD 9604 protocols.
Acquisition quality is the foundation of reproducible results. When sourcing AOD 9604, prioritize suppliers that provide third-party HPLC purity certificates (greater than 99%), mass spectrometry confirmation of the correct molecular weight, endotoxin testing results (LAL assay), and sterility documentation for injectable-grade material. Preclinical hype versus clinical reality is a well-documented pattern in peptide research, and it often starts with inconsistent sourcing rather than flawed study design.
AOD 9604 holds GRAS (Generally Recognized as Safe) status for food applications in some jurisdictions, but this does not extend to injectable research use. Regulatory status varies significantly by country, and researchers must verify local rules before acquisition or use in any protocol.
Protocol design checklist for AOD 9604 research:
- Select the appropriate delivery route (subcutaneous is most common for in vivo; oral for GI absorption studies)
- Establish baseline metabolic markers before treatment, including adipokine panels and β3-AR expression
- Choose controls that match your model’s receptor profile
- Define primary endpoints that are mechanistically relevant, not just body weight
- Plan for at least two time points to capture both acute and chronic effects
- Document reconstitution method, storage conditions, and lot number for every experiment
Use the AOD 9604 dosing calculator to convert preclinical benchmarks into species-appropriate working concentrations. This step is frequently skipped and is one of the most common sources of protocol error.
Pro Tip: When using the 2mg standard, write a formal dose rationale document that references the specific preclinical or clinical data supporting your chosen concentration. This protects your methodology during peer review and makes your negative findings publishable rather than dismissible.
Expert perspective: Why AOD 9604 is more than preclinical hype (and what researchers really need to know)
With the practical guidance in place, let’s step back and reflect on the deeper lessons AOD 9604 offers to scientific inquiry.
Most research guides on AOD 9604 treat the failed Phase IIb trial as a footnote. We think it’s the headline. The peptide’s story is a near-perfect case study in what happens when species-specific receptor biology is underweighted in translational planning. The rodent data was never wrong. It was just answering a different question than the human trials were asking.
The variables that matter most, β3-AR density, adipose tissue composition, delivery route, and subject metabolic phenotype, are routinely glossed over in peptide literature summaries. Researchers who engage with those variables seriously will extract more value from AOD 9604 protocols than those chasing the headline efficacy numbers.
There’s also a transparency problem in the field. Negative findings from peptide studies rarely get published, which means the research community keeps rediscovering the same dead ends. If your AOD 9604 protocol produces null results, document them rigorously and share them. That data has real scientific value. The field advances faster when researchers treat inconclusive outcomes as findings rather than failures.
Ready to advance your AOD 9604 research?
If you’re preparing to use AOD 9604 in your next protocol, here’s how to ensure robust results and easy sourcing.
The Peppy&Me research platform provides secure, verified access to research-grade peptides including AOD 9604 2mg, with full documentation for purity, sterility, and sequence confirmation. Researchers can use the platform’s dose calculation tools to streamline protocol setup and reduce pre-experiment preparation time.
For institutional teams or labs requiring consistent supply, private label peptides are available through the partnership program, supporting both small-scale research runs and larger longitudinal studies. Every product is backed by third-party testing and designed to meet the documentation standards your protocols require.
Frequently asked questions
What is AOD 9604 2mg and how is it different from GH Fragment 176-191?
AOD 9604 is a synthetic peptide fragment (Tyr-hGH 177-191) stabilized with a tyrosine modification and disulfide bridge, offering improved proteolytic stability and more consistent receptor binding compared to the unmodified GH Fragment 176-191.
What did animal studies reveal about AOD 9604’s efficacy?
Obese Zucker rats receiving 500 mcg/kg/day showed a 50% reduction in weight gain, but these results reflect rodent-specific β3-AR distribution and do not directly predict human outcomes.
Are there safety concerns with AOD 9604 in human research?
No IGF-1 elevation, glucose intolerance, or GH-like effects were observed across six clinical trials involving approximately 900 subjects, establishing a strong tolerability profile for research use.
How should researchers determine dosing for AOD 9604 protocols?
Dosing should be grounded in published Phase IIa benchmarks and scaled appropriately for the model used; researchers should document all rationale clearly to support reproducibility and peer review.