How to Buy Cagrilintide: Secure Sourcing Guide

TL;DR:
- Proper preparation and documentation are essential for safe, legal research with cagrilintide.
- Verifying supplier quality through independent testing and transparency prevents contamination and inconsistency.
- long-term research requires building trust and communication with vendors for reliable peptide supply.
Cagrilintide is generating serious attention across research circles, biohacking communities, and health-focused entrepreneurial ventures. It’s a long-acting amylin analog with a growing body of clinical evidence behind it, particularly in the context of metabolic and weight research. But the growing interest has created a crowded, unregulated market where finding a reliable, high-quality source is far harder than it should be. Phase 3 trial data underscores that individual access to cagrilintide for research is an entirely different category than clinical program enrollment. This guide gives you a structured, safety-first approach to sourcing it correctly.
Table of Contents
- What you need before buying cagrilintide
- Step-by-step guide to sourcing cagrilintide
- Compliance and safety in cagrilintide purchases
- Verifying the quality and results of your purchase
- What most buyers overlook about sourcing cagrilintide
- Upgrade your sourcing with Peppy&Me
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Preparation is crucial | Gather legal documentation and understand the risks before purchasing cagrilintide. |
| Source from verified vendors | Compare reputable suppliers and insist on quality assurance for any peptide purchase. |
| Compliance matters | Follow up-to-date regulations and safety measures to avoid penalties or unsafe products. |
| Verify every batch | Always check your peptide’s purity and effectiveness through lab testing and clinical data. |
What you need before buying cagrilintide
Before you place a single order, preparation is everything. Jumping straight to a supplier without the right documentation, knowledge, and infrastructure in place is one of the most common mistakes researchers and entrepreneurs make in this space. Getting this foundation right will protect both your research integrity and your legal standing.
Legal and research documentation
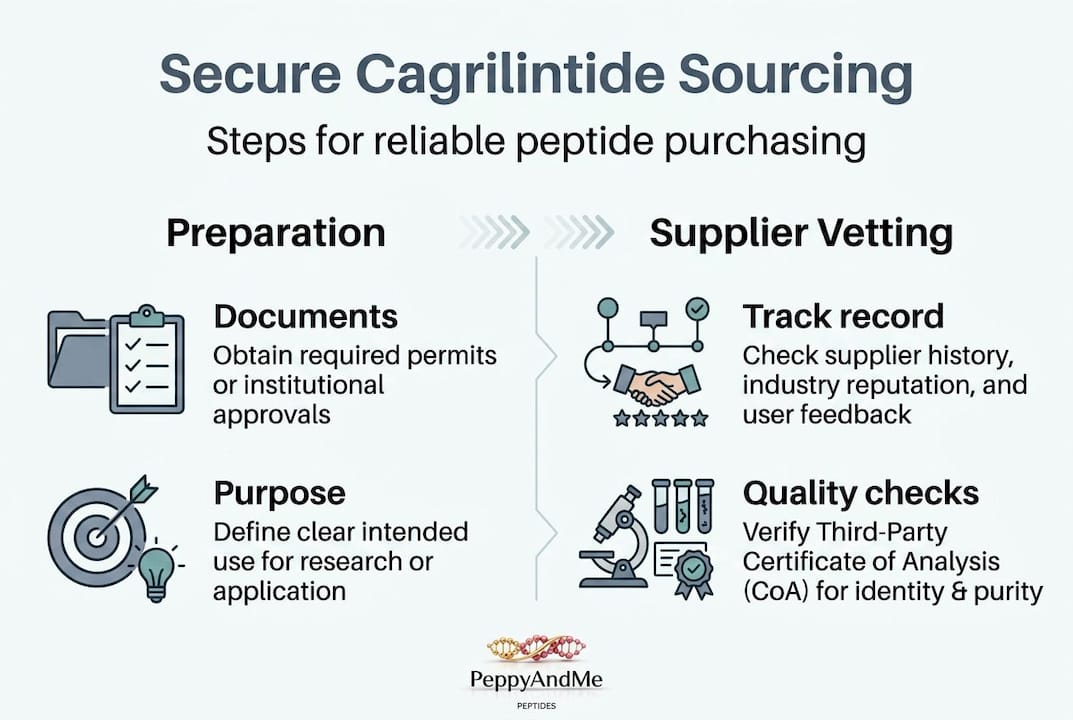
The first item on your checklist is documentation. If you’re affiliated with a research institution, confirm that your institution’s protocols cover the use of research peptides, including novel amylin analogs like cagrilintide. Institutional review boards and biosafety committees often have specific requirements for handling bioactive compounds. Independent researchers and biohackers operating outside formal academic settings should understand the regulatory environment in their jurisdiction before purchasing.
Keep a written record of your intended research purpose. This doesn’t need to be a full scientific protocol, but it should clearly state the research objectives, expected methods, and how the peptide will be handled and stored. This documentation protects you and helps you engage seriously with reputable suppliers who ask the right questions.
Understanding clinical vs. research use
Cagrilintide’s efficacy and safety profile have been established through rigorous clinical trials, but that research context is entirely separate from the research peptide market. Clinical-grade cagrilintide is manufactured under pharmaceutical GMP (Good Manufacturing Practice) standards with strict regulatory oversight. Research-grade peptides exist in a different category, intended for laboratory use only and not for human therapeutic application.
This distinction matters for two reasons. First, it shapes what you can legally do with the product. Second, it affects what quality standards you should expect and demand from your supplier. Research-grade does not have to mean low quality. It simply means the use context differs from FDA-approved pharmaceutical products.
Equipment and certifications often needed
Before your cagrilintide arrives, ensure your lab or workspace is equipped for safe handling:
- Sterile reconstitution supplies: Bacteriostatic water, syringes, and sterile vials
- Cold storage: Most peptides degrade rapidly without proper refrigeration at 2 to 8 degrees Celsius
- PPE: Gloves, lab coat, and eye protection for handling bioactive compounds
- Accurate measurement tools: Digital milligram scales and calibrated syringes
- Chain of custody records: Logbooks or digital records tracking receipt, reconstitution, and use
Pro Tip: Before sourcing cagrilintide, review the latest Research Peptides Blog for handling protocols and storage guidelines that reflect current best practices. Small preparation mistakes at this stage can compromise your entire research workflow.
Experienced buyers also spend time vetting potential sources before committing to a purchase. Look for suppliers that publish certificates of analysis, clearly state their testing methodologies, and have verifiable third-party testing relationships. A supplier who cannot or will not answer detailed questions about their testing practices is a red flag.
Step-by-step guide to sourcing cagrilintide
After gathering your prerequisites, proceed to the actual process of selecting and purchasing cagrilintide. This isn’t a one-step process. It’s a structured evaluation that separates reliable sources from risky ones.

1. Define your specification requirements
Start by writing down exactly what you need: quantity, concentration, purity threshold (most serious researchers require 98% or higher), and any specific formulation requirements such as lyophilized powder vs. solution. Having this written out before you contact suppliers prevents scope creep and makes supplier comparison much cleaner.
2. Research candidate suppliers
Search for suppliers with a documented track record in research peptides. Look for published reviews from verified researchers, active presence in professional forums, and transparent disclosure of their testing practices. The laboratory sourcing insights available from established platforms can help you identify quality benchmarks to compare against.
3. Request documentation before ordering
Contact each candidate supplier and ask for:
- Certificates of Analysis (CoAs) for recent cagrilintide batches
- Third-party lab testing reports, not just in-house results
- Documentation of endotoxin testing, sterility testing, and heavy metals screening
- Lot and batch traceability information
A reputable supplier provides this information without hesitation. Reluctance or vague responses to these requests is a serious red flag.
4. Compare domestic vs. international vendors
| Factor | Domestic Suppliers | International Suppliers |
|---|---|---|
| Shipping speed | Faster, often same-day | Typically 7 to 21 days |
| Regulatory clarity | Clearer jurisdiction | Variable and complex |
| Import risk | Minimal | Customs delays possible |
| Pricing | Often slightly higher | Sometimes lower but variable |
| Quality consistency | Easier to verify | Harder to audit remotely |
Domestic suppliers generally offer more accountability and faster recourse if something goes wrong. International vendors can sometimes offer lower prices, but the tradeoffs in reliability and regulatory clarity often don’t justify the savings for serious research programs.
5. Identify deal-breakers before committing
“Not all products labeled as cagrilintide match the standards or dosage tested in clinical trials.” This is one of the most critical facts in the research peptide market and one that too many buyers overlook until a problem surfaces.
Watch for these warning signs: no published CoAs, prices that seem unrealistically low, no customer support contact, vague or missing storage instructions, and no clear return or replacement policy for out-of-spec products.
Pro Tip: Ask every potential supplier to confirm whether their cagrilintide is tested by an independent, accredited third-party laboratory. If the answer is “we do all testing in-house,” consider that a yellow flag at minimum. Independent verification removes conflicts of interest that can compromise data integrity.
Compliance and safety in cagrilintide purchases
Once you’ve chosen a supplier, address compliance and safety to avoid costly or dangerous mistakes. This phase is where many otherwise well-prepared buyers stumble, often because they underestimate the operational discipline required.
Research vs. clinical distinction in regulatory terms
The clinical and research procurement difference has direct regulatory consequences. Clinical-grade cagrilintide is subject to FDA oversight and is dispensed only through licensed pharmacies and healthcare providers. Research-grade cagrilintide purchased from a peptide supplier operates under different rules, typically governed by the intended use classification and the supplier’s compliance with applicable chemical and laboratory standards.
Misrepresenting your intended use when purchasing research peptides is a legal and ethical violation. Always be transparent about your research context.
Safety standards to demand from every supplier
At minimum, your cagrilintide source should meet these standards:
- Purity: 98% or higher, verified by HPLC (high-performance liquid chromatography)
- Mass accuracy: Confirmed by mass spectrometry to verify peptide identity
- Sterility: Tested to confirm no microbial contamination
- Endotoxin levels: Tested via LAL (limulus amebocyte lysate) assay to rule out bacterial byproducts
- Heavy metals: Screened to eliminate contamination risk from synthesis byproducts
“The difference between clinical use and research peptide procurement has safety and regulatory consequences that go beyond paperwork.”
Lab and storage safety for bioactive peptides
Once your cagrilintide arrives, proper storage is not optional. Lyophilized (freeze-dried) peptides should be stored at minus 20 degrees Celsius for long-term stability. After reconstitution with bacteriostatic water, keep the solution refrigerated between 2 and 8 degrees Celsius and use it within 4 to 6 weeks. Avoid repeated freeze-thaw cycles, which degrade peptide structure and reduce potency.
Keep detailed records of every reconstitution event, including date, volume, concentration, and who handled the product. This creates an auditable trail that supports research integrity.
Pro Tip: Stay current by bookmarking trusted sources for guidelines on peptide regulations, as regulatory guidance in the research peptide space can shift with minimal public notice. Setting up alerts for relevant regulatory agency announcements keeps you ahead of compliance changes.
Ethical pitfalls to avoid
Never purchase cagrilintide with the intention of reselling it for human use, sharing it informally with individuals outside your research program, or representing it as pharmaceutical-grade to others. These actions carry serious legal risk and undermine the credibility of the broader research peptide community.
Verifying the quality and results of your purchase
After securing and storing your cagrilintide, it’s critical to verify its quality and monitor outcomes for safety and efficacy across your research work. Many buyers treat receipt of the product as the finish line. In reality, it’s the starting point of quality assurance.
Step-by-step verification checklist upon receipt
- Inspect the packaging: Check for intact seals, proper labeling with lot and batch numbers, and no signs of moisture or temperature compromise during shipping
- Cross-reference the CoA: Confirm the CoA included with your shipment matches the lot number on the vial
- Request independent verification: For high-stakes research, send a sample to a third-party analytical lab for HPLC and mass spec confirmation before using the product
- Check concentration accuracy: Compare the stated concentration against your expected measurements when reconstituting
- Document everything: Record the condition of the product upon arrival, including photos if anything looks unusual
Comparing lab analysis with clinical data
Published clinical trial outcomes for cagrilintide provide a useful benchmark for understanding expected peptide behavior. When comparing your supplier’s batch data against these standards, focus on peptide purity and molecular weight as the two primary markers of authenticity.
| Quality Metric | Research Standard | What to Watch For |
|---|---|---|
| Purity (HPLC) | 98% or higher | Values below 95% indicate quality issues |
| Mass accuracy | Within 1 Dalton of target | Significant deviation suggests wrong peptide |
| Endotoxin level | Below 1 EU/mg | High endotoxin levels indicate contamination |
| Sterility | No microbial growth | Any positive result is disqualifying |
| Moisture content | Below 8% | High moisture accelerates degradation |
Monitoring for expected vs. unexpected results
In research settings, monitoring begins as soon as any experimental use is initiated. Cross-reference your observations with the known safety and activity profile from peer-reviewed literature. Cagrilintide’s behavior in controlled research contexts should align with published data when the product is authentic and properly handled.
Batch inconsistencies are a real issue in the research peptide market. If you notice variability between batches, from the same supplier or across different suppliers, document it carefully. Bring this data back to your supplier and request a formal response. A reputable vendor will investigate and offer replacement or remediation. Suppliers who dismiss quality concerns or delay responses should be removed from your approved vendor list. For additional authenticity testing advice, established research platforms provide updated guidance on validation methods.
What most buyers overlook about sourcing cagrilintide
Here’s what years in the research peptide space make clear: the buyers who struggle most are not the ones who skip due diligence on purpose. They’re the ones who assume a single vendor check is enough to guarantee long-term supply consistency.
Trusting a certificate of analysis at face value is risky, even from suppliers with good reputations. CoAs can reflect a single batch that passed testing while other batches in the same run had issues. The only protection is independent, routine verification across every order, not just the first.
Consistency is also far harder to achieve than most guides acknowledge. Cagrilintide is a structurally complex peptide, and synthesis quality can vary with changes in raw materials, synthesis equipment, or even personnel. Long-term research programs that depend on batch-to-batch reproducibility need to build vendor relationships that include supply chain transparency and advance notification of any manufacturing changes.
The most effective sourcing strategy is collaborative, not transactional. Build genuine communication channels with your vendors. Ask about their manufacturing partners, update schedules for quality testing, and policies for notifying customers of batch changes. Platforms that support deep-dive peptide sourcing with educational resources tend to attract suppliers who take these standards seriously, making them valuable starting points for identifying trustworthy partners.
Upgrade your sourcing with Peppy&Me
Sourcing cagrilintide with confidence requires more than just finding a vendor. It demands a platform built around transparency, verified quality, and research support.
Peppy&Me is designed for exactly this. Every peptide is third-party tested for purity, sterility, endotoxin levels, mass accuracy, and heavy metals, with full lot and batch traceability from manufacturer to warehouse. For entrepreneurs ready to scale, the peptide sourcing partnerships program offers private label solutions backed by Peppy&Me’s operational infrastructure. Use the built-in dose calculator tool for precise measurement support, and explore the full product range, including options like peptide for relaxation, all within a secure, membership-based platform built for professionals.
Frequently asked questions
Is it legal to buy cagrilintide for research in the US?
Purchasing cagrilintide for legitimate research purposes is generally permitted, though clinical use is subject to strict FDA regulations and is separate from research peptide procurement. Always verify local and federal guidelines before ordering.
How can I verify the purity of research peptide cagrilintide?
Request a certificate of analysis from your supplier and confirm results with independent third-party lab testing, since unregulated research products may not match pharmaceutical-grade consistency. HPLC and mass spectrometry are the gold-standard verification methods.
What are the main side effects associated with cagrilintide?
Gastrointestinal symptoms are the most commonly reported adverse effects, and GI adverse events were documented in up to 72.5% of participants in clinical trials. Nausea is the most frequent individual complaint in published data.
Are research peptides like cagrilintide the same as pharmaceutical-grade products?
No. Research peptides lack the regulatory oversight of pharmaceutical products, and not all labeled cagrilintide matches the purity or dosage standards from clinical trials. Third-party testing and documented quality standards are essential safeguards when sourcing research-grade peptides.